Limitations in how we
currently measure success for
patients with Fabry disease
Limitations in how we
currently measure success for
patients with Fabry disease
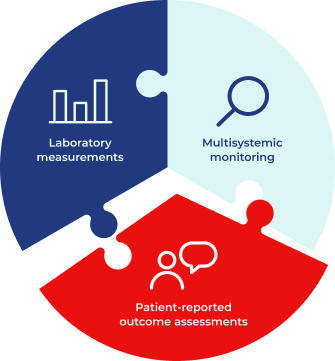
A range of assessments and schedules are recommended to monitor different organ systems in adult patients with Fabry disease.1 However, there has been no systematic way to evaluate the frequency and impact of signs and symptoms, or to identify which are the most debilitating and most important in terms of quality of life (QoL).2
Current recommendations include:
-
Laboratory measurements, including Gb3 levels, lyso-Gb3 levels, albuminuria test and eGFR.1 Additional monitoring dictated by each patients individual clinical course3
-
Multisystemic monitoring – all relevant systems must be assessed, treated and monitored individually4
Can patient-reported outcomes offer a different perspective?
Patient reported outcomes may be measured with:
-
SF-36 questionnaire: assesses 8 domains of QoL - Physical Functioning, Role Physical, Bodily Pain, General Health, Vitality/Energy, Social Functioning, Role Emotional, and Mental5
-
EQ-5D questionnaire: assesses 5 domains of QoL - mobility, self-care, usual activities, pain, and anxiety/depression5
Better understanding of QoL in different disease states and improved understanding of the influence of specific symptoms and complications on QoL may facilitate targeted treatment, and improve the well-being of Fabry patients.6
THOROUGHLY UNDERSTANDING PATIENT EXPERIENCES IS KEY IN MEASURING THE BURDEN OF FABRY.2
eGFR, estimated glomerular filtration rate; Gb3, globotriaosylceramide; lyso-Gb3, globotriaosylsphingosine; SF-36, 36-Item Short Form Health Survey.
Improving multidisciplinary care for patients, now and in the future
Clinical evaluations should involve a multidisciplinary team of subspecialists, which should be coordinated by a physician experienced in the care of patients with Fabry disease.3
A care team may include physicians of the following specialties:1
Cardiology
Nephrology
Neurology
Psychology
Genetics
Primary care
If you or your patients don’t have access to all of the specialists noted above, directing your patients to the following resources may help them learn more about their condition:
ESC
European Society of Cardiology
EKPF
European Kidney Patients Federation
EFNA
European Federation of Neurological Associations
EFPA
European Foundation for Psychologists and Analysts
EGAN
Patients Network for Medical Research and Health
EURORDIS
The Voice of Rare Disease Patients in Europe
Helping your patients navigate their care team
Supportive care is important.7 To help your patients understand the importance of a multidisciplinary approach, consider advising them that:
Therapeutic management of Fabry disease requires a multidisciplinary approach by medical specialists experienced in treating this rare condition.4
Comprehensive monitoring regardless of age, sex, or treatment status, should be conducted at regular intervals.4
Annual evaluations ensure that they receive optimal interventions and support.3
Results from each specialist will come together to form a complete picture of their health.1
Working with specialists early will allow for the collection of baseline measurements, which can help the team see signs of progression early.1

It’s important for them to work with a multidisciplinary care team, even if they are not currently experiencing symptoms in a given part of their body.1,5
Their family members with Fabry may experience the disease in a distinct way.2
Introducing Chiesi Global Rare Diseases
At Chiesi Global Rare Diseases, we’re working to build a brighter future for patients.
We want to revolutionize the lives of patients living with rare diseases by providing an integrated set of definitive solutions.8
The patients are the beginning and the end of our journey. For them, we work in close partnership with patients, caregivers, patient associations, healthcare practitioners, and regulatory and pricing authorities.8
We strive to build a brighter future for the patients we serve; we relentlessly pursue sustainable growth with passion, courage, teamwork, and innovation.8
We are fully committed to rethinking what is possible in Fabry disease. Together, we can take action to make a change.
References:
- Ortiz A, et al. Mol Genet Metab. 2018;123(4):416-427.
- Hamed A, et al. Orphanet J Rare Dis. 2021;16:285.
- Desnick RJ, et al. Ann Intern Med. 2003;138(4):338-346.
- Eng CM, et al. Genet Med. 2006;8(9):539-548.
- Arends M, et al. Orphanet J Rare Dis. 2015;10(77):1-10.
- Arends M, et al. J Inherit Metab Dis. 2018;41(1):141-149.
- Germain DP. Fabry disease. Orphanet J Rare Dis. 2010;5:30.
- Chiesi Global Rare Diseases Institutional Presentation, June 2021.
NAVŠTIVTE
Pro pacienty - V případě potřeby hlásit nežádoucí účinek léku se obraťte na svého lékaře a požádejte ho, aby vyplnil a předložil příslušnou kazuistiku příslušnému zdravotnickému úřadu v souladu s požadavky farmakovigilance platnými ve vaší zemi. Nicméně mějte prosím na paměti, že každý pacient může hlásit jakékoli takové případy přímo národnímu systému hlášení.: CHIESI CZ s. r. o.- Smrčkova 2485/4, 180 00 Praha 8 - Česká republika. Tel.: +420 261 211 850. E-mail: safety.cz@chiesi.com
Pro zdravotnický personál: Pokud potřebujete ohlásit zjištěný nežádoucí účinek léku, obraťte se na příslušný zdravotnický ústav v souladu s požadavky stanovenými legislativou o kontrole léčiv: CHIESI CZ s. r. o.- Smrčkova 2485/4, 180 00 Praha 8 - Česká republika. Tel.: +420 261 211 850. E-mail: safety.cz@chiesi.com
Nyní opouštíte webové stránky RethinkFabry.cz.
Webové stránky, na které vstupujete, společnost RethinkFabry.cz nevlastní ani je nespravuje.
Tyto stránky jsou určeny odborným pracovníkům ve zdravotnictví. Informace nejsou určeny pro laickou veřejnost. Pro laickou veřejnost je určena samostatná sekce pro pacienty.
Potvrzuji, že jsem odborníkem ve smyslu §2a Zákona č. 40/1995 Sb., o regulaci reklamy, ve znění pozdějších předpisů čili osobou oprávněnou předepisovat léčivé přípravky nebo osobou oprávněnou léčivé přípravky vydávat. Beru na vědomí, že informace obsažené dále na těchto stránkách nejsou určeny laické veřejnosti, nýbrž zdravotnickým odborníkům, a to se všemi riziky a důsledky z toho plynoucími pro laickou veřejnost.
